Content Sections
On 8 June this year, the Food Safety Authority of Ireland (FSAI) issued a 160-page report advocating “new upper limits for vitamins and minerals to safeguard health”.
Download full FSAI report
ANH-Intl’s analysis of the report (summarised below) suggests that the new limits could threaten the public’s access to beneficial levels of some vitamins and minerals, notably niacin, vitamin B6, folate, vitamin B12, vitamin D and zinc. Such restrictions may violate fundamental rights and freedoms of the Irish people.
The FSAI, the Irish food regulator, says it will be working with the food supplement industry to establish guidance for the marketing of safe vitamin and mineral supplements in Ireland. However, ANH-Intl argues that imposing limits on maximum levels that are below the upper levels may be disproportionate. Among the reasons for this is that upper levels do not adequately take into account differences between risk/benefit profile of different forms (e.g. pyridoxine and pyridoxamine, vitamin B6; folic acid and reduced 5’-methyltetrahydrofolate, folate). Nor do they account sufficiently for differences in response or requirement between individuals.
Why has the Irish regulator issued upper levels for vitamins and minerals?
The EU Food Supplements Directive (2002/46/EC) has always intended to implement EU-wide, HARMonised limits on minimum and maximum levels of vitamins and minerals used in food supplements (Article 5). However, the European Commission, after years of deliberation on the subject with EU Member State authorities, stakeholders, NGOs and the European Parliament, has decided for the time being to not proceed with issuing harmonised levels. This has led to something of a Wild West, with some Member States issuing, or at least planning to issue, their own maximum levels.
In the publication of this 2018 report, the Irish authority has taken the first step in making clear it will use a standard risk assessment approach that can then lead to regulatory enforcement. In no uncertain terms, the FSAI indicates its intention to remove from the Irish market any food supplements it deems as unsafe.
What is a Tolerable Upper Level or TUL?
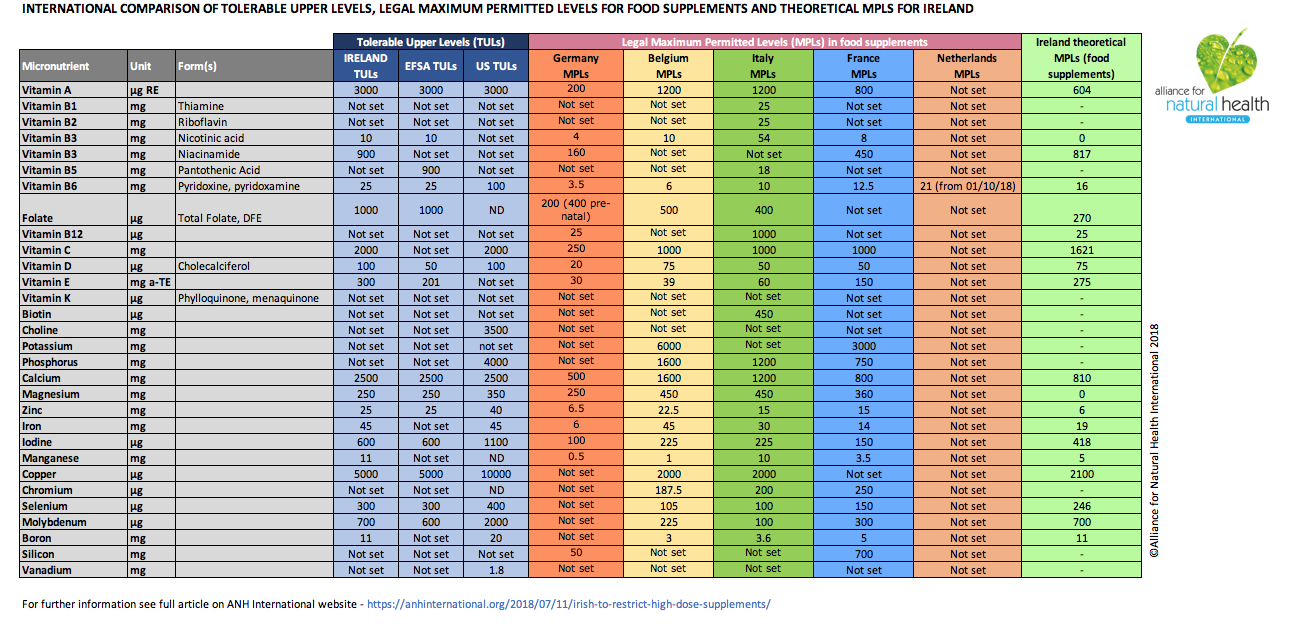
The FSAI report specifies tolerable upper intake levels (TULs) for 21 of the 30 vitamins and minerals permitted in food supplements in Ireland. These are defined as “the highest level of long-term daily intake of a nutrient, from all sources, judged to be unlikely to pose a risk of adverse health effects to humans.”
The levels proposed in the FSAI report coincide approximately or exactly with those of the European Food Safety Authority (EFSA) set in 2006 or the US Institute of Medicine (IoM) for the majority of vitamins and minerals, and differ for a few (see Table 1 below). These differences are claimed to be intended to account for the specific needs and “safety” of the Irish population.
It should be noted that TULs are determined on the basis of the nutrient form viewed as triggering the most sensitive adverse effect among the most susceptible group of individuals. They do not take into account the needs of those who have above-average requirements as they ignore any consideration of benefit.
It is something of an irony that EFSA’s 2006 report was presided on by Prof Albert Flynn of University College, Cork in Ireland, who was the then chair of EFSA’s Panel on Nutrition, Novel Foods and Food Allergens (NDA) (up until 2012). Prof Flynn, as it happens, is also chair of the Scientific Committee of the FSAI that has issued the 2018 report.
ANH-Intl determinations of theoretical Irish maximum permitted levels (MPLs)
FSAI indicates in its report that the food supplement industry should apply a “standard risk assessment approach for evaluating the safety of vitamins and minerals in food supplements in Ireland. This is on the basis that the daily amount of a micronutrient from a food supplement, added to the usual daily intake from food sources, i.e. from foods, including fortified foods, but excluding supplements, in the highest consumers, i.e. the 95th percentile, should not exceed the tolerable upper intake levels (UL) for the population group(s) for whom the food supplement is intended.”
We have therefore calculated ‘theoretical’ maximum permitted levels or MPLs using this approach. Quite simply, we subtracted the amounts consumed by the highest Irish consumers (i.e. the 95th percentile) from the FSAI’s TUL. If the resulting figure was negative, we made it zero. We have also taken into account prescription only medicines (POM) legislation mentioned in the FSAI report (SI No. 540/2003) which specifies dosages at which certain vitamins become medicinal. Where these levels are lower than those determined with the above calculation, we have had to use the maximum daily dose above which the dosage would be deemed as medicinal.
For the sake of simplicity, we have only performed calculations for adults, excluding pregnant or lactating women.
TABLE 1 [click on table image to enlarge and open as a PDF]
ANH-Intl determinations of regulatory impacts on micronutrient benefit
A major problem linked to a health authority limiting access to higher dose nutrients in supplements relates to the negative impacts on those who have higher than average needs. Such restrictions can limit an individual’s ability to stay healthy or restore their health.
If the restriction of public choice is without adequate scientific basis, the restriction may be considered a violation of fundamental rights and freedoms, such as under Article 8 of the European Convention on Human Rights. It is our view that if a vitamin or mineral form that is permitted for sale in Ireland is present in a food supplement at a dose in which that nutrient form cannot be regarded as unsafe, any restriction of its sale on the part of the national authority should be illegal. Obviously, it would be a European Court, probably the European Court of Justice rather than the European Court of Human Rights, that would be required to make such a judgment.
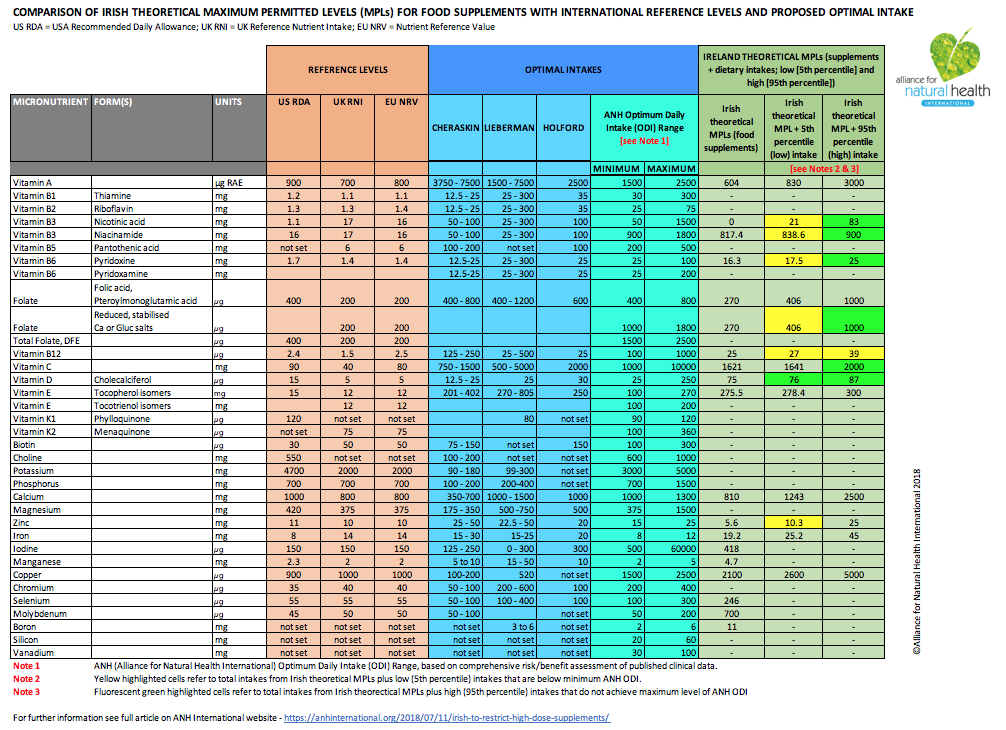
In this regard, we have determined that if the Irish government was to proceed with its proposed approach, restrictions on 5 vitamins and one mineral may be illegal. These are:
- Niacin (as nicotinic acid)
- Vitamin B6 (especially in the pyridoxamine and pyridoxal forms)
- Folate (especially in the reduced, 5’-methyltetrahydrofolate forms)
- Vitamin B12 (all forms)
- Vitamin D (all forms)
- Zinc (all forms)
The determinations on which these conclusions are based are summarised in Table 2.
In brief, the figures shown in the two right-hand columns of Table 2 reflect total intake of consumers who derive the lowest intakes from their diet, and those who derive the highest intakes from their diets, respectively, with the addition of intakes based on the theoretical MPLs.
We have then compared these levels with various estimates of optimal intake. We have used four different sources:
- Optimal intake for vitamin/mineral supplements as proposed by Dr Emanuel Cheraskin
- Optimal supplementary intakes proposed by Shari Lieberman in her 2007 book, Real Vitamin and Mineral Book: The Definitive Guide to Designing Your Personal Supplement Program
- Patrick Holford’s Optimum Daily Allowances
- ANH-Intl’s Optimum Daily Intake (ODI), based on risk/benefit assessment of published clinical data.
TABLE 2 [click on table image to enlarge and open as a PDF]
Where to from here?
We’re in the process of taking these and related issues up with the Irish health trade. In our view this trade is the unsung hero in the healthcare equation in Ireland. Often vilified for selling supplements that are claimed as harmful or ineffective based on a misrepresentation of the science. Yet offering a gamut of healthy foods, herbal teas, botanicals and a host of other ingredients that help keep so many people, who understand their value, healthy.
It beggars belief that Professor Albert Flynn and colleagues will hand this distorted and over-simplistic view of micronutrient safety to enforcers in the FSAI. These enforcers, unaware of the implications of their brief, will then go on to limit public access to food supplements that have decades of demonstrated safe sale on the Irish market.
Should disproportionate enforcement commence, we are convinced it will not be accepted by the Irish people lying down. And we will be standing firmly at the side of those wishing to defend higher dose supplements that can be shown to be safe.
Alongside their safety, these supplements also help large numbers in Ireland to maintain their health. Many consider that spending money on natural health products is preferable to paying the typical €50 fee per visit to a general practitioner (GP).
Watch this space.
Read Irish Times article 'New guidelines on vitamins and mineral supplement consumption published' published Tue, Jun 5, 2018, 19:57
Back to Health Choice campaign








Comments
your voice counts
12 July 2018 at 11:10 am
There is just one very glaring problem with vitamin supplements?... They create healthy people! And healthy people compete directly with Medicine, and Pharmaceutical Drug Industry! That medicine with all of its poisonous side effects, and insane death statistics.
If Professor Flynn has a real reason he wants to control vitamins, then he should start out by very clearly stating why? Since it is common knowledge around the world that vitamins are almost completely harmless!
Professor Flynn and colleagues could really help everyone by putting their attention where it belongs? In the Pharmaceutical Drug Industry that is causing so much physical and mental suffering, addiction and death worldwide!... Because the people in Ireland, and everywhere else in this world have no problems using their own health-promoting vitamins.
12 July 2018 at 11:12 am
The 'harmonisation' of vitamins and minerals across Europe has been going on for decades, I first wrote to my MP when we had the ridiculous empty lorries with slogans across the sides being driven across Europe heralding 'safer' supplementation!
And yet governments are quite happy to flood our drinking water with toxic fluoride and add folic acid to our bread whether we like it or not!
People should be able to choose what they want to put in their bodies, education is the key.
12 July 2018 at 12:05 pm
I absolutely agree with your article, this is just another step on the road to Totalitarian government , inch by inch. Show us the adverse effects of any of these, then decide if these controls or better directions on use are necessary. We loose the freedom of choice to our own personal health, while the HSE cannot even reduce the 700,000 waiting on hospital waiting lists. The next step will be mandatory vaccinations. Too many people are beginning to look after their own health via vitamins and minerals, and this does not suit the directed course of the unelected EU Commission, who no doubt have hidden interests. Doctors know little to nothing on Nutrition they would rather prescribe a pharmaceutical, but once the Pharmaceutical Companies control the vitamin and mineral market, then they will have absolute control. It would be interesting to see Prof Flynn's interests and qualifications.
Ireland is a silly little nation, we have to first with everything that is of a controlling nature, and last in matters of human benefit, just to appear progressive in the dictatorial EU. Thank for your article
12 July 2018 at 12:32 pm
no way should they cut down the rdi of vitamins.big pharma wants to shut them down so they can sell their poisonous drugs that kill thousands of people each year with impunity, like opiods, prescription drugs etc.they are the ones that should be controled. i v selenium, lipo spheric vit c, etc at high doses saved people from there death beds within one week.check 60 mins.
12 July 2018 at 1:31 pm
Quite apart from the evil anti life pattern that emerges whether consciously intended or not in whole or in parts, I raise the issue of the 'dumb user' or consumer, who is protected from an education so as to 'need' protecting from a world that they are neither equipped nor trusted to (understand). They are thus utilities running upon utilities in a largely dumb or top-down managed system. They become units in a system of utility - where such 'usefulness' is essentially determined by profit and power of the rule makers.
While narrative assertion has always over simplified the nature of our world, our self and our relations, systemic narrative control as a manipulative set of shocks or limits to human 'systems' whereby to induce compliance and conformity to normalised systemic violence is of a different order - as the 'outsourcing' (giving up) of a sovereign will to a system of imposed rules of 'broad spectrum dominance'. This term is often applied to military power but it refers to so called soft power and power via vectors of stealth and guile. Medical need, response and interventions are one such vector.
Underneath the 'power' to actually set and implement such a global agenda is a drive to both escape fears by outsourcing personal and shared protection to powers that usurp or take responsibility away instead of serving its ongoing development as an educative support. And thus also to give power to a fantasy of control over 'chaos' where chaos is assigned to any threat to the system and to the privileged status of those who identify in justifying such fantasies of control in terms of saving us from a greater 'chaos'. As such the system itself is operating chaos as a system of rules divorced from a true relational being.
The "foxes are running the hen-house" is not merely a matter of malevolent or misguided 'persons' but of the very currency of our thought.
Just as our financial 'currency' is corrupted from a true grounding in shared worth, so is our vocabulary in its accepted usage.
As I see it, we are being forced (by our own errors) to have to re-evaluate everything we believed and acted true - where the nature of such belief runs beneath our current sense of consciousness because they constitute the 'reality' we have adapted to and are conformed or framed within.
Much of our 'healthcare' has been dedicated to maintaining a personal sense of out-of-true 'freedom' against the sense of 'authority' as an imposing structure on our will. And so an oppositional 'will' seems to promise freedom from a fear (belief) that truth demands sacrifice (of self). In this do we DO the very thing we fear and mis-take to be true - while operating under the conviction we are protecting against evil done to us or threatening us.
True authority is our highest discernment or indeed deepest self-honesty. But while willingly but unmindfully subject to the false, the partial, and the mask of the well-intentioned, we give unto 'Caesar' what is due unto 'God' - and let 'God' serve as the placeholder for true relation within our self, between ourselves and the embracing of our existence in a movement of being that resonates the true will or desire of living, that a mind-in-wish subverts or usurps to personal or private agenda.
Aligning in health is aligning in wholeness of being. This is an embracing living Perspective, not a dividing, ruling out attempt to rule over.
There are avenues to restoring of presence and function that are 'ruled out' by the exclusively 'physical' model and mapping of life. yet the exploration of the 'physical' is revealing a vibrational field of resonant frequency 'communication' that life simply 'is and does'. And so some of the effect of being increasingly denied support in the frame of our 'current knowing', will be to drive the expansion of knowing through the willingness to 'step outside' the model. This is always a call for deeper self-honesty, and willingness of true desire - for without it we simply meet and reinforce the fear-bounding of the human 'conditioning'.
True desire is not in the head but moves to our recognition when we 'be still and know'.
The pain of the (fear-belief in) loss of the heart's knowing seeks in a world of external searching, whose substitutes cannot heal or make whole - and yet if such are our terms and conditions, Life can only meet us in what we are willing to accept.
Thankyou for the text box for a reply. The acting from willingness for wholeness of being grows willingness in the mind of all as one. The wilfulness of parts seeking 'personal' wholeness at expense of each other and of the whole - and therefore themselves, is a mind that knows not what it does by virtue of its masking persona or self-blinding 'narrative identity'.
12 July 2018 at 2:57 pm
MPL is expressed as mg/day?
12 July 2018 at 5:10 pm
Hi David
In the table, the MPL is expressed as mg/day or mcg/day, but always represents the maximum permitted daily intake from supplements. The Tolerable Upper Levels (TULs) are from all sources. Please let us know if this isn't what you were asking.
Best wishes
Meleni
Your voice counts
We welcome your comments and are very interested in your point of view, but we ask that you keep them relevant to the article, that they be civil and without commercial links. All comments are moderated prior to being published. We reserve the right to edit or not publish comments that we consider abusive or offensive.
There is extra content here from a third party provider. You will be unable to see this content unless you agree to allow Content Cookies. Cookie Preferences