By Robert Verkerk PhD
Executive and scientific director, ANH-Intl
Energy is everything. Without enough of it, important internal functions as well as physical activity is limited. But the ramifications of energy deficiency go well beyond the typical list of mitochondrial diseases that are well-known to medicine; it contributes directly or indirectly to all diseases, especially chronic and degenerative disorders, ranging from chronic fatigue syndrome and fibromyalgia, to neuropathic pain syndromes, dementia and Alzheimer’s disease, through to obesity, diabetes and even heart failure and various cancers. In short, a lack of energy within the system is often the reason why the tremendously intelligent self-regulatory or self-healing systems within our bodies become compromised.

“Illuminating the Energy Spectrum: Exploring the evidence and emerging clinical solutions for managing pain, fatigue, and cognitive dysfunction”, was the theme for this year’s annual international conference for the Institute for Functional Medicine, held in Dallas, TX, held between 30 May and 1 June 2013.
Our executive coordinator, Meleni Aldridge, and I made our annual pilgrimage to the IFM conference as we have for the last 9 years in my case, and 8 in Meleni’s.
This piece aims to bring back, in layperson’s terms, what we feel are some of the most important ‘take homes’ and ‘clinical pearls’ – ones that have the ability to be life changing among those who suffer from sub-optimal energy production.

There were no more seats available at this year’s Institute of Functional Medicine annual conference, with 1200 participants present – the largest attendance to date
Energy fundamentals
- Chemical energy in our bodies is ultimately derived from the sun’s fusion energy. This energy conversion that provides the fuel to run the myriad of reactions and processes within our bodies occurs through the action of mitochondria within our cells. These are tiny organelles, typically just 0.5 to 1.0 micrometer (μm) in diameter, that owe their evolutionary origin to ocean-dwelling bacteria.
- Mitochondria provide the hub for cellular respiration that produce energy in the form of adenosine triphosphate (ATP) from glucose. Essentially, mitochondria take the oxygen emitted by plants (the ‘exhaust gas’ of photosynthesis), as well as nutrients from our food, including glucose and various vitamin cofactors, and convert them to chemical energy in the form of ATP. While most cells in the body are dependent on ATP as their energy source, brain cells are unique in their reliance on glucose as their primary energy source.
- Mitochondria rely on two distinct but interrelated mechanisms for energy production, the Krebs or citric acid cycle, that occurs in the matrix (interior region) of the mitochondria, and oxidative phosphorylation (also known as the ‘electron transport chain’), which occurs in the mitochondrial membrane. The ATP yield from oxidative phosphorylation is considerably greater than that from the Krebs cycle, although the Krebs cycle produces the electrons required to initiate the chain of redox reactions associated with oxidative phosphorylation.
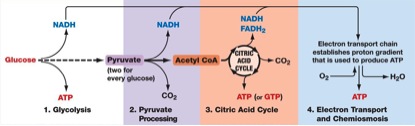
Key steps in cellular respiration: from glucose to ATP, via the Krebs cycle and the electron transport chain
- Mitochondria have numerous other functions other than energy (ATP) production, including the regulation of cellular growth and metabolism, signalling, cell death (apoptosis) and important functions in calcium homeostasis.
- The number of mitochondria and the amount of energy produced by an individual mitochondrion can vary greatly from cell to cell, and from person to person.
- Mitochondrial dysfunction is strongly linked to ageing and neurological disorders such as Parkinson’s disease, dementia and Alzheimer’s disease.
- Ketogenic diets, caloric restriction and intake of specific nutrients can help to significantly improve mitochondrial energy output.
- Improving our mitochondrial reserve may be among the most effective ways of avoiding premature ageing.
- Energy within our bodies flows through interrelated networks, rather than the simplistic pathways typically shown in textbooks.
Energy dysfunction and disease
- ‘Tired all the time’ or TATT represents one of the most common reasons why people visit their doctor. Most doctors fail to provide interventions that are helpful through a lack of recognition of the symptoms of mitochondrial dysfunction, in turn brought about through inadequate functional medicine training.
- ‘Leaky gut’ or hyperpermeability of the gut lining is another very common condition, although its prevalence is often inadequately recognised by mainstream doctors. The increased permeability appears to be at least partially caused by mitochondrial dysfunction, which prevent the proper function of ‘tight junctions’ through the action of zonulin in the mucosal lining of the gut. Altered zonulin response also affects the permeability of the blood-brain barrier. This means that ingested or metabolic toxins or pathogens are able to migrate through the gut lining into the body, precipitating a range of possible problems, including for cells distant from the gut, such as the brain. Altered zonulin response caused by mitochondrial dysfunction is now thought to be an important mechanism for autoimmune conditions such as coeliac disease caused by gluten.
- A number of cell types are particularly vulnerable to sub-optimal ATP production by mitochondria. These cells, that typically contain the greatest number of mitochondria — up to 60% by volume — could be viewed as ‘yellow canaries’. The cell types, as well as the conditions arising in them when suffering energy deficit, are: neurons, leading to cognitive impairment; cardiocytes, leading to cardiometabolic impairment; myocytes, leading to various myalgias such as fibromyalgia and chronic fatigue syndrome, and; enterocytes, leading to leaky gut and various autoimmune diseases. While energy deficit may not be the sole cause of these disorders, given that genetic susceptibility plays a role in some, energy deficit is always implicated to some extent and improving energy production may often be the most important clinical intervention. While prevention is always better than cure, it is always best to recognise symptoms of energy deficit as early as possible and provide interventions before it causes serious disease.
- A wide range of mitochondrial toxins abound and exert their effects primarily by inhibiting or uncoupling oxidative phosphorylation. Such toxins include antiretrovirals used to treat HIV/AIDS, many antibiotics (remember: mitochondria were originally bacteria), statins (causing statin myalgia or myopathy) and the now ubiquitous persistent organic pollutants (POPs). Post-menopausal women may be among the most vulnerable to mitochondrial toxins because their reduced oestrogen is unable to protect against the uncoupling of the electron transport chain.
- There is an increasingly clear relationship between mitochondrial dysfunction and obesity, mediated through the different origins, functions and gene expression associated with white, beige and brown adipocytes (fat cells). White adipocytes, with low mitochondrial complements, are the typical hallmark of obesity, while brown adipocytes, with high mitochondrial concentrations, are associated with heat production (thermogenesis) and protection from cold. It has recently been found that a new type of beige adipocytes can occur within white fat depots and can be activated to release energy in the form of ATP by specific chemical signals and in response to exercise and caloric restriction. Beige adipocytes can differentiate to either white or brown adipocytes, and are susceptible to macrophage attack following up-regulation of the immune system when visceral adipose tissue (VAT) has turned ‘angry’, in turn triggering the activation of the renin-angiotensin-aldosterone system (RAAS) associated with hypertension and type 2 diabetes. This leads to a common scenario experienced by obese, overweight and diabetic patients, in which they complain that they are unable to lose weight irrespective of what they eat. The beige fat needs to be trained to become brown, high energy-releasing, thermogenic fat.
- The loss of muscle mass with age (sarcopenia) is an important morbidity, often dramatically reducing quality of life in older people and increasing healthcare costs. Recent research in rhesus monkeys has shown that ‘metabolic sarcopenia’ may be triggered by a shift in energy metabolism caused by oxidative effects within mitochondria.
- Mitochondrial dysfunction may occur as a result of biotransformation associated with xenobiotics such as pesticides. Plant secondary metabolites such as resveratrol from red grape skins and curcumin from turmeric may protect against mitochondrial damage by such xenobiotics.
‘Clinical pearls’
Among the most important mechanisms for increasing mitochondrial concentration and activity are:
- High caloric intake and sedentary lifestyles cause an increase in production of advanced glycation end products (AGEs). Glycated proteins in turn not only increase diabetes, heart disease and cancer risk, they also cause increased rate of shrinkage of the hippocampus leading to memory loss and neurodegenerative diseases.
- Exercise intolerance can be a symptom of mitochondrial dysfunction or disease.
- Low fat diets are generally associated with high (often simple) carbohydrate diets, and are a major cause of recent increased rates of obesity, diabetes and neurodegenerative diseases in Western societies.
- Probably the most powerful mediator for increased mitochondrial biogenesis (new formation) and activity is exercise. Exercise regularly (on at least 4 days/week, totalling at least 7 hours/week), and include short bouts of intensive exercise, such as high intensity interval training (HIIT).
- Ketogenic (high healthy fat, low carbohydrate) diets are strongly associated with neuroprotective effects through its effect on increasing cellular concentrations of, and energy outputs by, mitochondria. Healthy fats include coconut oil, rich in medium-chain triglycerides (MCTs), unfiltered extra virgin olive oil, nut oils and avocado oil.
- Caloric/dietary restriction. This should include regular overnight (12-16 hour) fasts, preferably with exercise at the end of the fast, where the first meal of the day (e.g. breakfast or lunch) is consumed within 30 minutes of completion of exercise.
- Every day, consume plenty of vegetables and to a lesser extent fruits (aim for no more than 3 portions a day, but only 2 if you have sugar handling issues) from all six colour groups (blue/purple, red, orange, white/tan, green and yellow) in order to provide diverse phytonutrients including those that protect against mitochondrial damage and excessive oxidation.
- A range of 10 key opinion leaders in the functional medicine arena, including ANH-Intl executive coordinator, Meleni Aldridge, were interviewed on their views regarding specific nutrition and lifestyle interventions for healthy mitochondria, aging and energy production. Out of the information gathered from the 10 interviews, a new IFM 'Healthy Aging Core Food Plan' has been created. In summary, the top 12 super foods for eating and drinking your way to healthier mitochondrial function are: avocado, spinach, seaweed, pomegranate, blueberries (all berries), broccoli (and all cruciferous vegetables), grass-fed buffalo/beef, wild Alaskan salmon, almonds, coconut oil (virgin, organic), olive oil (cold pressed, unfiltered/cloudy, extra virgin), green tea. Mangoes almost made it on to the list, and would certainly have done so if the list were not limited to 12!
- Specific nutrients that have been demonstrated to be highly protective of mitochondria while preventing inflammasome activation include: resveratrol (derived from red grape skin or Japanese knotweed), curcumin (from turmeric root), quercetin (from citrus peel) and cruciferous vegetables (kale, broccoli, cabbage, etc), and stewed apples with added cinnamon.
- B vitamins and coenzyme Q10 (notably the reduced form, ubiquinol) are among the key cofactors required for energy production via the Krebs cycle and electron transport chain.

Contributors to the IFM’s "Healthy Aging Core Food Plan”,from left to right: Dr Kristi Hughes, Dr Terry Wahls, Michael Ash DO, Meleni Aldridge BSc, Dr Catherine Willner, Lyra Heller MA, Dr David Perlmutter, Barb Schiltz RN, Deanna Minnich PhD, Dr Mark Houston. Dr Stephen Sinatra, was also a contributor, but absent.
Memorable quotes from this year’s IFM symposium
David Rakel MD, physician, researcher and author of Integrative Medicine, explained that, “Habituated neuro-networks of comfort are so hard to get out of — and they’re a good reason why so many doctors have difficulty believing the stuff they didn’t learn at med school.” Emphasising the importance of empathy in a clinician’s approach to his or her patients, Dr Rakel provided research evidence that showed that “empathy has been trained out at medical schools.” He added, “Bureaucracy expands to meet the needs of the expanding bureaucracy”.
Neurologist David Permutter MD is a strong protagonist of ketogenic, grain-free diets and exercise as the primary means of preventing or even reversing neurodegenerative diseases such as Alzheimer’s disease. In his opening lecture, he told the 1200-strong audience of clinicians, scientists and researchers that; “While the research into ketogenic diets might have started around 1921, humans first went on ketogenic diets around 2.6 million years ago.” Emphasising the point further, he said, “There is no diet than can do better than a ketogenic dietary change.”
Jeff Bland PhD emphasised that complex systems such as rainforests tended to be stable systems, while simples ones, such as a cornfield, were unstable. This analogy could be applied to energy networks in human beings, as well as their optimisation by dietary and lifestyle means. Resilience is increasingly being seen as the ultimate marker of an individual’s health. Commenting on this, Dr Bland said, “Response of the expression of your genes to your environment gives you your resilience to a changing environment”. He added, “Medicine for the individual is emerging, while medicine for the average is gone.”
Dr Bland stressed the difference between different types of fat in the body, contrasting especially ‘angry fat’ from what he referred to as “ho-ho-ho Santa Claus” friendly fat. Adding to this, he suggested that one big pizza meal with sodas (including glycated proteins, unhealthy fats and high sugar content) will “turn on the inflammasome” in the gut.
Rollin McCraty PhD, executive vice president and director of research of the Heart Math Institute, defined resilience as “The capacity to prepare for, recover from, and adapt in the face of stress, challenge or adversity.”
Anthropologist and shaman, Alberto Villoldo PhD, founder of the Four Winds Society reminded participants that over the majority of the 2.6 millions years of human existence, humans were not so much hunter/gatherers, but scavenger/gatherers. He said that hunting was always difficult and we ate what we could and had to endure long periods of fasting between meals. Summing up, he opined: "A bunch of guys would go out and find some roadkill, clean it up, then come back and say 'hey honey look what I've caught for us to eat, can we have sex now?” He added that during fasting, the insulin/glucose control system was turned off and that we had to go into the ketogenic state to fuel the brain. Which is why we're so well adapted to fasting rather than feasting!
Call to Action
- Take responsibility for your health, and do what you can to enhance your body’s innate energy production systems by following the advice given in the Clinical Pearls section above.
- Get the message out as far and wide as you can using social media, word-of-mouth and other means.
- If you need help or an individualised plan, seek advice from a practitioner trained in functional medicine.








Comments
your voice counts
06 June 2013 at 12:53 am
v informative article. Seaweeds are a wonderful food , but if a person is inland there must be inland equivalents - samphire is another superfood I would add tho very regional. I think every region has its own superfoods indigenous to the region and often free for foraging like the old scavenging ways when we gathered as we found..hedgerows give a lot in this way and woodlands - mushrooms , nettles, dandelions some easy finds for the novice and berries :) We dont have avacado indigenous to my country ( Ireland) but I think a real dairy full fat butter can provide great benefit - esp for those aging with gum issues or inflammation from mercury fillings - really butter or ghee will heal inflammation , side affliction of mitochondria dysfunction..no?
It should not be a problem for the vegetarian as no animal is slaughtered..:)Green leafy vegetables actually supply alpha linolenic fat acids along with flax seed and hemp seed - also walnuts. For happiness add the Brazil nut!
Your voice counts
We welcome your comments and are very interested in your point of view, but we ask that you keep them relevant to the article, that they be civil and without commercial links. All comments are moderated prior to being published. We reserve the right to edit or not publish comments that we consider abusive or offensive.
There is extra content here from a third party provider. You will be unable to see this content unless you agree to allow Content Cookies. Cookie Preferences