Content Sections
The Federal Institute for Risk Assessment (Bundesinstitut für Risikobewertung [BfR]) published a paper earlier this month in the Journal of Consumer Protection and Food Safety that sets the scene for how it proposes the German government should handle maximum levels of vitamins and minerals in food supplements and fortified foods. Germany, like other EU countries, is taking the issue into its own hands in the knowledge that the European Commission has parked its plans to harmonise these levels EU-wide.
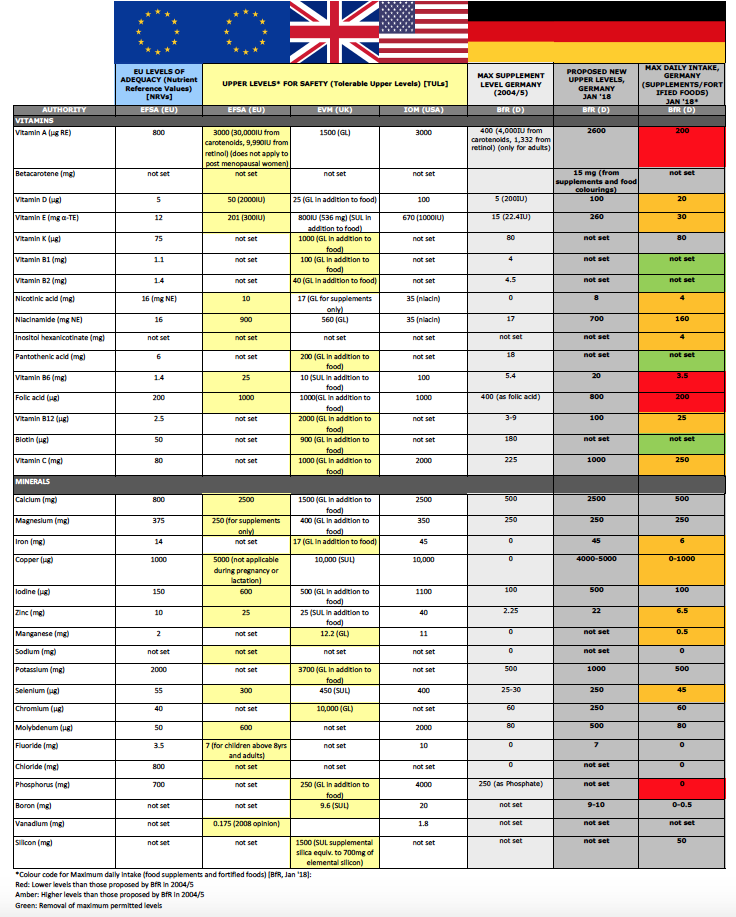
The paper, published in the German language with an English abstract, continues to rely on a scientifically flawed approach to risk assessment that we comprehensively exposed in the peer-reviewed journal Toxicology in 2010. The results of the current analysis, based on a greater body of available research, has resulted in some wins and losses in its maximum daily recommendations compared with its previous reports from 2004 and 2005.
‘Winners’
It’s hard to have real winners when the BfR is using a scientifically flawed model. But there are at least some vitamins that have proposed maximum levels that are greater than those set by the BfR in 2004. We’ve given these a green traffic light rating (see Table below).
Vitamin B1 (thiamin), vitamin B2 (riboflavin), pantothenic acid (B5) and biotin no longer have maximum permitted amounts in recognition of their extreme safety and water solubility.
Also, moving in the right direction, compared with the 2004/5 BfR reports, were proposed increases in maximum daily amounts for vitamin D, niacin (as nicotinic acid, niacinamide and inositol hexanicotinate), vitamin B12, vitamin C, vitamin E, iron, copper, zinc, manganese and selenium (see Table below). In many cases, these increases are trivial so as to make no real difference physiologically – and certainly not therapeutically (a notion that is entirely prohibited by EU and German authorities).
However, in many cases these increases are nothing like those that should have occurred had the BfR followed a more scientific approach. As we have also demonstrated in another journal article in Toxicology in 2010, the scientific approach should be one based on risk/benefit assessment. Such an approach would result in levels that don't discriminate against the majority of the population using an overly precautious approach centred around the most sensitive members of the population.
Losers
The BfR’s latest report stays with its previously highly restrictive maximums for vitamin K, calcium, magnesium, iodine, potassium, chromium and molybdenum.
Astonishingly, the BfR fails again to take into account the effect of micronutrient form, and so applies the risk associated with iron sulphate to iron bisglycinate, the risk of magnesium oxide to magnesium citrate, and so on. Given the outpouring of research on the subject, it ignores any differences between the risk/benefit profile of vitamin K1 (phylloquinone) and K2 (menaquinone).
Table: Comparing reference levels of micronutrients in the EU against those of Germany from 2004/5 and 2018 respectively
Unscientific, disproportionate and unjust
The latest BfR report shows once again how Germany is prepared to go against the flow of both the science and the law. The European Commission (EC) has in the past successfully challenged national authorities (e.g Commission v Austria, Case C-150/00) for disproportionately restricting vitamins selling freely and safely in other EU Member States and so interfering with the function of the single EU market. If companies don't try to defend their rights to free movement of goods, a right that Germany is required to respect as a result of its obligation under the Treaty on the Functioning of the European Union, the EC may need to strike again.
Germany may well need to face the courts in the future and is far from guaranteed a win.
At ANH-Intl, we are continuing our work on risk/benefit analysis and are expecting to see the results of this work used initially in national courts within the EU later this year.








Comments
your voice counts
25 January 2018 at 6:56 pm
Dear Rob - this is really in response to your introductory message on the weekly newsletter that included this article.
I am glad to see you coming out of the closet - in a helpful way - and of course in the light of unfolding events I am not surprised.
'Coming Out’ is a term usually assigned to a socially stigmatized gender orientation - but in the sense I feel it - goes to the nature of our true witness instead of complicity in the false.
If we don't come out for sovereign will as an embodiment of it - then we assent to exchanging our humanity for a management of lies - or if you prefer illusions passing off as a fake life, a fake world of fake relationships.
My sense is that blindness is easier to maintain in the dark - and that by extending the qualities of life in what we do and say - we shine the witness and reminder to those who have sought safety in hiding and become adapted to the dark. There is another way of looking at this (whatever we believed it to be).
But in case you are not already aware, the study of human hopes and fears as a vector of control for social/political engineering is no less embedded in technological development - and largely drives and shapes it.
A true foundation is more profound that a reaction to a wrong, regardless how grievous the wrong.
Our responsibility to wake up to where we are ‘hackable’ or ‘phishable’, so as to restore wholeness and health of a conscious discernment, is being obligated by a destructive agenda and outcome that is active behind the masks of institutional capture as it is beneath the masking of social mores.
I wont ramble on - but I wanted to honour what you are doing and offer perspectives without which good intentions miss the underlying nature of the disease.
The technocratic replacement for life is not founded on scientific expertise. I believe you have recognized this. However - insofar as bureaucratic systems can be held to account, and you can - please do.
Joining in shared purpose - as an expression of a cultural inspiration is different from a fear defined reaction. But I believe the need is to extend a true witness of aligned purpose and not to be framed and defined in reaction.
Health choices and life choices are an order of cause and effect that reaches down into our creative being - and not merely a management system for those who wield control of it.
Your voice counts
We welcome your comments and are very interested in your point of view, but we ask that you keep them relevant to the article, that they be civil and without commercial links. All comments are moderated prior to being published. We reserve the right to edit or not publish comments that we consider abusive or offensive.
There is extra content here from a third party provider. You will be unable to see this content unless you agree to allow Content Cookies. Cookie Preferences